FDA Investigation
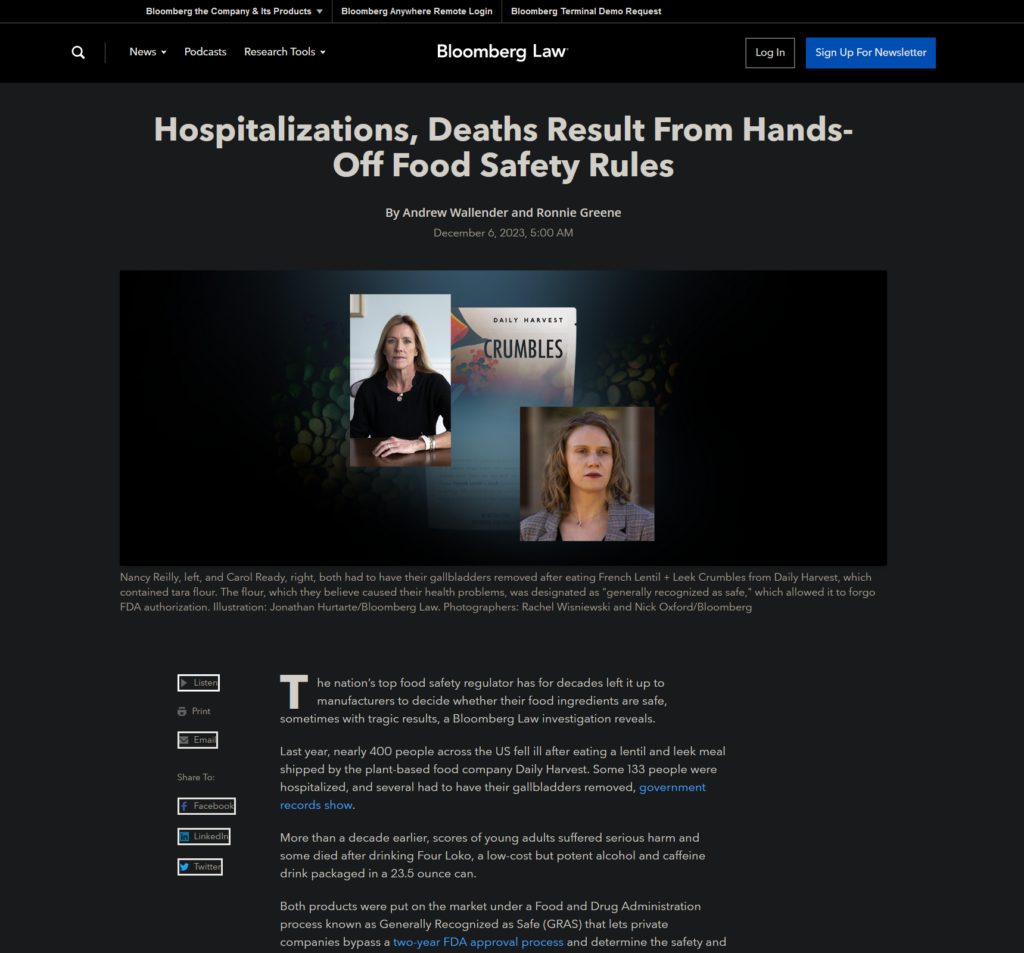
This story came about through my reporting on the toxic “forever chemicals” known as PFAS. Several sources mentioned that types of PFAS were potentially making their way into the food supply because manufacturers were self-identifying the substances as “generally recognized as safe,” therefore bypassing any initial U.S. Food and Drug Administration oversight.
My colleague and I decided to dig into the “generally recognized as safe,” or GRAS, process to investigate the impact of allowing manufacturers to self-designate their products as safe. We uncovered a system that leaves consumers vulnerable to harm and obfuscates information on what Americans are eating.
The reporting process involved interviews, FOIA requests, reading through dozens of lawsuits, and an analysis of a voluntary program that allows manufacturers to disclose to the FDA substances that they determine to be GRAS. To analyze the voluntary GRAS disclosures, I used R to scrape and further analyze the FDA’s database.
Graphic from the story
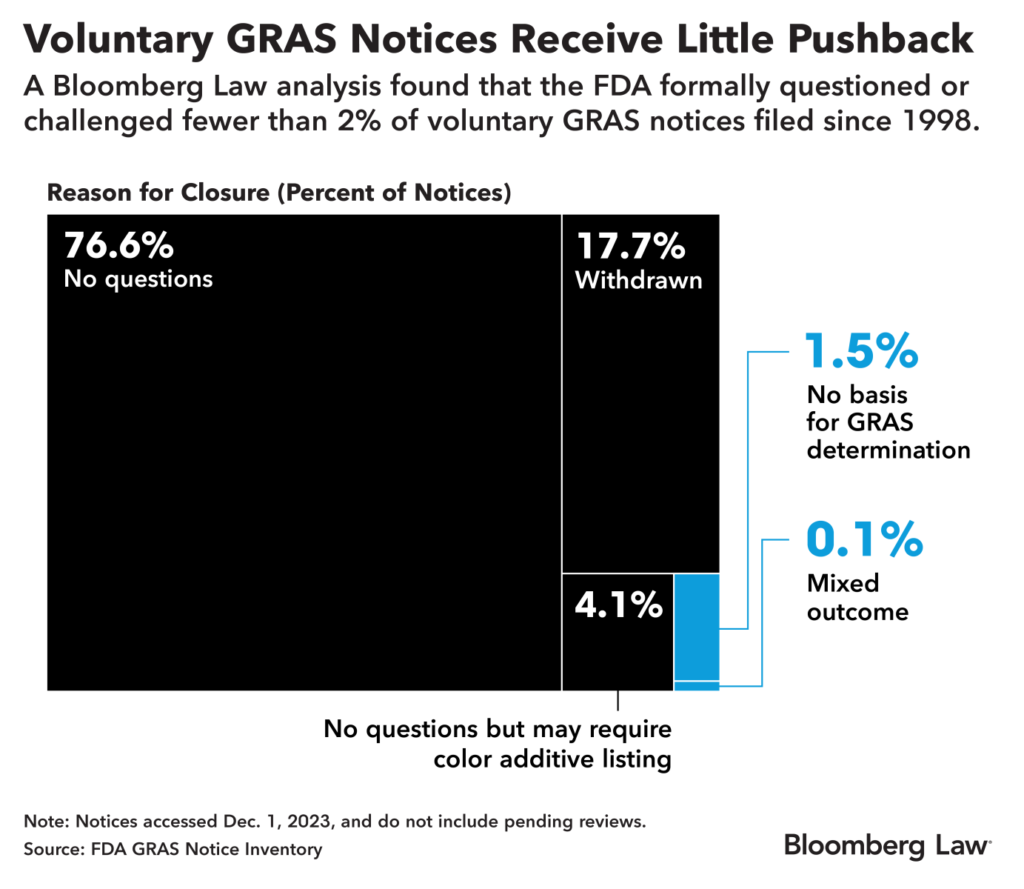
Description
Bloomberg Industry Group
December 6, 2023
Investigation into how the U.S. Food and Drug Administration regulates the safety of food ingredients. The story uses first-hand experiences, FOIA'd documents, and an analysis of available data to reveal that food oversight is often outsourced to private industry.